Part 1 in The Endocrine Chronicles
What’s Leptin?
Discovery
Leptin, named from the Greek leptos, or “thin”, was discovered in 1994 following a decades-long effort to better understand the role of the endocrine system in weight regulation. Beginning in the 40s and 50s, some physiologists began to question the prevailing notion that weight was entirely controlled through personal decisions to eat less and move more, suggesting instead that body weight is closely regulated by the endocrine system. Disruption in homeostasis, rather than simply willpower, might be a cause of excess weight gain.
In 1953, Gordon Kennedy proposed the “lipostat theory,” a hypothesis that an individual’s fat mass signals the brain via the hypothalamus, alerting the endocrine feedback loop of how much energy intake might be needed.1 This theory gained support in 1959, when review of an early 40s study confirmed that hypothalamus lesions could cause obesity in rats. Researchers speculated that the lesions were preventing the brain from absorbing a signal of satiety.
A 1949 experiment sought a genetic explanation when a group of laboratory mice offspring became unexpectedly obese, despite having normal-weight parents. A mutation of a gene on chromosome 6 was eventually mapped; the gene was dubbed the obese (ob) gene. Ob-ob homozygous mice had the mutation and became obese. Researchers determined that the ob gene likely coded for a hormonal signal that would inform the hypothalamus of satiety, and the mutation of ob-ob individuals interfered with this pathway.1

Decades later, Jeffrey Friedman and colleagues finally managed to clone the ob gene and demonstrate that it was expressed in adipose tissue, coding for a hormone that informed the hypothalamus of fat mass status and satiety. Leptin’s official discovery confirmed the importance of the endocrine system for regulation of body weight and eating behavior.
How Leptin Works
Leptin is:
- A hormone, one of the body’s many chemical messengers
- A cytokine, a group of small proteins/peptides that signal through surface receptors but do not cross the cell membrane
- An adipokine, a family of hormones secreted by adipose (fat) tissue
- Anorexigenic, or a hormone related to appetite suppression
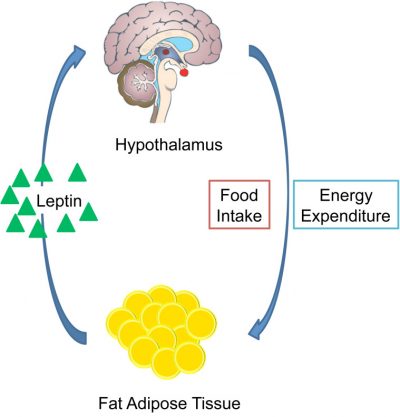
In humans, the lep gene (corresponding to ob) is located on chromosome 7. Leptin is produced and secreted directly from adipose tissue. The hormone acts directly on leptin cell receptors in the hypothalamus to communicate energy storage status. Leptin’s primary function is the negative feedback loop of energy stores and satiety. Fasting decreases leptin levels, resulting in hunger cues, while feeding increases levels, signaling satiety.2
What Can Go Wrong: Leptin Resistance
Simple serum levels of leptin do not represent the whole story of endocrine weight regulation, and the picture is less clear for humans than the original ob/ob lab mice. As expected, low leptin levels were found in about 5 to 10 percent of obese humans but many overweight individuals actually have higher serum leptin than their average weight counterparts.1
Since leptin’s 1994 discovery, both obese humans and ob/ob mice have been tested with leptin injections. Although these were helpful to a small percentage of subjects compared to the control, the vast majority did not respond, leading researchers to believe that leptin deficiency was not the primary issue, but leptin resistance. 2 Serum leptin was found in normal or above average amounts, but subjects were nonresponsive or underresponsive to it. The phenomenon may be similar to the role of insulin resistance in metabolic syndrome. The causal relationship between leptin resistance and obesity is still unclear.2
Why Leptin Matters for Astronauts: Space Changes
Circadian Fluctuations
It’s common for human hormone secretion to fluctuate throughout a 24 hour cycle. Leptin is no exception. This “circadian network is synchronized via neuronal and hormonal signals by a master clock located in the suprachiasmatic nuclei of the hypothalamus, mainly entrained to the environment by light perceived by the retina.”3
In other words, hormonal fluctuation – including that of leptin – is heavily influenced by light exposure. Changing the timing of exposure to light can alter this “circadian clock,” throwing off the normal rhythm of leptin secretions. Leptin, released based on a 24 hour cycle, has been shown to rise in correspondence to normal sleeping hours. This makes sense, as decreased leptin in the middle of the night could cause humans to wake up from hunger.
Melatonin, known as a sleep-inducing hormone, is primarily made at night and secreted by the pineal gland in response to increasing darkness. Release of melatonin also leads to secretion of more leptin at night.
Diurnal Cycles on Space Missions
A normal human diurnal cycle is one rotation of the Earth, or 24 hours. Astronauts, however, experience diurnal cycles of 90 minutes. In other words, space crew members experience a sunrise or sunset every 45 minutes.4 (Little Prince vibes, anyone?) Additionally, the low-light ambience on the ISS and spacecraft is also a concern, since the circadian clock is tied closely to light (or perceived light) exposure. As could be predicted by the melatonin connection, constant exposure to dimmer lights leads to continuously higher serum leptin levels and decreased appetite.
On the ISS, lights are kept at full brightness for 15.5 hours and dimmed for 8.5 hours in an effort to simulate the 24 hour cycle. However, lights are never fully dimmed in case of an emergency.7
Microgravity Effects
In contrast, extended exposure to a microgravity environment may have the opposite effect. In a study entitled “Food restriction and simulated microgravity: effects on bone and serum leptin,” Baek et. al. explored the possible effects of microgravity and reduced energy intake on possible bone loss and leptin levels in astronauts.5
48 Sprague-Dawley rats were placed into four groups:
- Regular cage activity + 100% baseline food intake (CC100)
- Regular cage activity + 70% baseline food intake (CC70)
- Hindlimb unloading via tail suspension* + 100% baseline food intake (HU100)
- Hindlimb unloading + 70% baseline food intake (HU70)
*Hindlimb unloading via tail suspension was meant to simulate a microgravity environment
After four weeks, all groups but the CC100 control had lost body weight and bone mass, the most drastic loss being in the HU70 group (microgravity simulation + calorie restriction).
The variables’ effects on leptin levels were significant. The control group remained stable, while the CC70 group experienced a 27% decrease and the HU100 group experienced a 60% decrease. Even more drastic, however, were the results for the HU70 group, where both variables were altered. Serum leptin levels were undetectable in these rats after 28 days. Testing larger volumes of serum did not change the results.5
The experiment showed that both reduced intake and microgravity simulation had a significant impact on leptin levels, but the hindlimb unloading much moreso. It has been hypothesized that leptin plays a role in building bone mass, although this may happen through a different pathway than the negative feedback loop of energy intake regulation.5 At first glance, it might seem as though a microgravity environment could counteract the leptin-increasing effects of continuous lower ambient light. However, lowering levels due to physical stress may have different ramifications.
Space Radiation
Most Earth-side radiation is lower-ionizing, like gamma rays and x-rays. They are referred to as low linear energy transfer (low-LET) radiation and cause less damage than the heavier-energy ions found in space.6 These ions include 12C, 16O, 28Si, and 56Fe. This high-LET radiation is a serious health concern in space missions, particularly those that may be long-term and outside low Earth orbit. Existing protection against these energy-heavy ions is inadequate.6
Current studies are concerned with high-LET radiation’s effect on the GI tract and incidence of colorectal cancer. In one study, Suman et. al. focused on 56Fe’s effect on adipokines, including leptin, and insulin-like growth factor 1 (IGF1) in the GI tract of mice. Leptin and IGF1 were specifically examined due to their positive correlation with GI illness, including IBS and colorectal cancer; this correlation is likely due to “increasing evidence that leptin/IGF1 signaling cooperates to promote cellular proliferation and carcinogenesis…”6
Two months after exposure to high-LET radiation, serum leptin levels were significantly higher than the control in the irradiated tissue samples. Additionally, leptin receptor expression and downstream signaling pathways were both increased.6 Researchers considered the ramifications of these findings for astronauts. On a trip to Mars, astronauts would likely get only one-tenth of the high-LET dose used in the study. However, exposure to 56Fe ions were still causing greater activity in the leptin/IFG1 signaling axis a year later, suggesting possible cumulative effects.6
Exposure to cosmic radiation, as shown by this high-LET study, causes an increase in serum leptin. This might contribute to the problem of reduced appetite and energy intake. Prolonged travel will only exacerbate the complications.
Countermeasures
When planning habitats for long-term travel and possible colonization, mitigation of altered leptin levels should be considered. There are several possibilities. Though not necessarily simple to execute, they could be vital to success of longer missions.
- Habitats that allow for regular circadian rhythms, where light exposure is closer to a 24-hour cycle rather than a 90-minute diurnal cycle
- In-space, on-site ability to produce anti-inflammatory animal proteins like salmon and other fish high in omega-3 fatty acids, as well as plant-based options like avocado, coconut, and olive oil. Continuous transport of animal proteins is impractical for close-to-home missions and impossible for extended ones. Growing fish on-site from embryos might be one option.4
- Pinealectomy, or surgical removal of the pineal gland, has been shown to reduce variations in clock genes responsible for adipokine secretion in rat subjects.3 While this is too extreme and likely damaging for humans, planning light and dark exposure times around the desired release time of melatonin from the pineal gland is one way to keep leptin levels normalized and lower during the day to stimulate appetite and energy intake.
- If light exposure is insufficient to regulate circadian leptin levels, or if maintaining a 24-hour diurnal rhythm is not always feasible, exposure to smaller, repeated meals might produce a similar effect. Rather than three large meals, offering six smaller meals at standardized times might produce a rhythmic secretion of leptin.3
Complexity of the endocrine system is one factor to consider in how human biology will react to long-term space travel and habituation. Circadian rhythms of hormones like leptin are easily thrown off when faced with altered diurnal cycles, space radiation, and inconsistent feeding. Leptin will be a central figure in the question of how to keep appetite consistent in space.
Sources
1. Li MD. Leptin and beyond: an odyssey to the central control of body weight. Yale J Biol Med. 2011 Mar;84(1):1-7. PMID: 21451778; PMCID: PMC3064240. / 2. Münzberg H, Morrison CD. Structure, production and signaling of leptin. Metabolism. 2015 Jan;64(1):13-23. doi: 10.1016/j.metabol.2014.09.010. Epub 2014 Sep 28. PMID: 25305050; PMCID: PMC4267896. / 3. Challet E. Circadian aspects of adipokine regulation in rodents. Best practice and research. Clinical endocrinology and metabolism, 2017, 31 (6), pp.573-582. 10.1016/j.beem.2017.09.003.hal-02557249. / 4. Tang et. al. Long-term Space Nutrition: A Scoping Review. Nutrients. 2022, 14 (194). https://doi.org/103390/nu14010194 / 5. Baek K, Barlow AA, Allen MR, Bloomfield SA. Food restriction and simulated microgravity: effects on bone and serum leptin. J Appl Physiol 104: 1086-1093, 2008. doi: 10.1152/japplphysiol.01209.2007. / 6. Suman S, Kumar S, Fornace A, Datta K. Space radiation exposure persistenty increased leptin and IGF1 in serum and activated leptin-IGF1 signaling axis in mouse intestine. Scientific Reports. doi: 10.1038/srep31853. / 7. “Sleeping with the Lights On.” Space Center Houston. November 19, 2019. Accessed August 5, 2022. https://spacecenter.org/sleeping-with-the-lights-on.
